Seznamy Neutral Atom Of Calcium Výborně
Seznamy Neutral Atom Of Calcium Výborně. A impartial calcium atom additionally has 20 electrons. What type of ion does calcium form? Which means in a impartial calcium atom, there are 20 protons in its nucleus. An ion is an atom of a chemical element that has an unequal number of electrons compared to protons. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2.
Nejlepší Atom Diagrams Electron Configurations Of The Elements
What type of ion does calcium form? An ion is an atom of a chemical element that has an unequal number of electrons compared to protons. 1s2 2s2 2p6 3s2 3p6 4s2.All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail.
How many protons are in a neutral atom of calcium? What type of ion does calcium form? Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). 1s2 2s2 2p6 3s2 3p6 4s2. How many protons are in a neutral atom of calcium? The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Which means in a impartial calcium atom, there are 20 protons in its nucleus. Remember that electrons are negative charges, and protons are positive charges.

How do you find the neutral atom?.. How many protons are in a neutral atom of calcium? 1s2 2s2 2p6 3s2 3p6 4s2. Calcium is a chemical element found in nature. Remember that electrons are negative charges, and protons are positive charges. 29.04.2021 · the electron configuration of a neutral calcium atom is : Atomic mass of calcium is 40.078 u. Click on to see full reply. What type of ion does calcium form?. An ion is an atom of a chemical element that has an unequal number of electrons compared to protons.

1s^2 2s^2 2p^6 3s^2 3p^6 4s^2.. . How many protons are in a neutral atom of calcium?

How do you find the neutral atom?. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The electron configuration of a impartial calcium atom is : 1s2 2s2 2p6 3s2 3p6 4s2. What type of ion does calcium form? Atomic mass of calcium is 40.078 u. A impartial calcium atom additionally has 20 electrons. The atomic mass is the mass of an atom. 21.11.2020 · atomic mass of calcium. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. 21.11.2020 · atomic mass of calcium.

Remember that electrons are negative charges, and protons are positive charges.. The electron configuration of a impartial calcium atom is : Now, in a "neutral atom", the number of protons must be. 25.10.2021 · the atomic variety of calcium is 20. Remember that electrons are negative charges, and protons are positive charges. All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail. A impartial calcium atom additionally has 20 electrons.. Which means in a impartial calcium atom, there are 20 protons in its nucleus.

A impartial calcium atom additionally has 20 electrons.. .. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2.
The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 29.04.2021 · the electron configuration of a neutral calcium atom is : The atomic mass is the mass of an atom. The electron configuration of a impartial calcium atom is : How many protons are in a neutral atom of calcium? An ion is an atom of a chemical element that has an unequal number of electrons compared to protons. Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail. Now, in a "neutral atom", the number of protons must be. Remember that electrons are negative charges, and protons are positive charges. Therefore, the number of electrons in neutral atom of calcium is 20.

Therefore, the number of electrons in neutral atom of calcium is 20... Therefore, the number of electrons in neutral atom of calcium is 20. In the case of the calcium ion, we have a calcium element with a positive charge of 2. 21.11.2020 · atomic mass of calcium. Atomic mass of calcium is 40.078 u. 29.04.2021 · the electron configuration of a neutral calcium atom is : The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.
:max_bytes(150000):strip_icc()/Calcium-58b602433df78cdcd83d4c16.jpg)
1s2 2s2 2p6 3s2 3p6 4s2.. Calcium is a chemical element found in nature. All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail. 1s2 2s2 2p6 3s2 3p6 4s2. Now, in a "neutral atom", the number of protons must be. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. How do you find the neutral atom?

Now, in a "neutral atom", the number of protons must be.. Atomic mass of calcium is 40.078 u. Remember that electrons are negative charges, and protons are positive charges. 21.11.2020 · atomic mass of calcium. Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). 29.04.2021 · the electron configuration of a neutral calcium atom is : A impartial calcium atom additionally has 20 electrons. Now, in a "neutral atom", the number of protons must be. Calcium is a chemical element found in nature. What type of ion does calcium form?. Therefore, the number of electrons in neutral atom of calcium is 20.

Remember that electrons are negative charges, and protons are positive charges. .. Which means in a impartial calcium atom, there are 20 protons in its nucleus.

Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). An ion is an atom of a chemical element that has an unequal number of electrons compared to protons. How do you find the neutral atom? 1s2 2s2 2p6 3s2 3p6 4s2. Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. 25.10.2021 · the atomic variety of calcium is 20. All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The electron configuration of a impartial calcium atom is :. The electron configuration of a impartial calcium atom is :

25.10.2021 · the atomic variety of calcium is 20.. Now, in a "neutral atom", the number of protons must be. Which means in a impartial calcium atom, there are 20 protons in its nucleus. How many protons are in a neutral atom of calcium? Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton)... In the case of the calcium ion, we have a calcium element with a positive charge of 2.

Click on to see full reply.. Atomic mass of calcium is 40.078 u. 1s2 2s2 2p6 3s2 3p6 4s2. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. Calcium is a chemical element found in nature. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 29.04.2021 · the electron configuration of a neutral calcium atom is : 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2.

Remember that electrons are negative charges, and protons are positive charges.. The electron configuration of a impartial calcium atom is : An ion is an atom of a chemical element that has an unequal number of electrons compared to protons. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Which means in a impartial calcium atom, there are 20 protons in its nucleus. How do you find the neutral atom? A impartial calcium atom additionally has 20 electrons. How do you find the neutral atom?

What type of ion does calcium form?.. Therefore, the number of electrons in neutral atom of calcium is 20. How do you find the neutral atom? Remember that electrons are negative charges, and protons are positive charges.. Now, in a "neutral atom", the number of protons must be.

Which means in a impartial calcium atom, there are 20 protons in its nucleus. All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail.. Now, in a "neutral atom", the number of protons must be.
21.11.2020 · atomic mass of calcium. 25.10.2021 · the atomic variety of calcium is 20. A impartial calcium atom additionally has 20 electrons... A impartial calcium atom additionally has 20 electrons.

29.04.2021 · the electron configuration of a neutral calcium atom is : 25.10.2021 · the atomic variety of calcium is 20.

Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). Therefore, the number of electrons in neutral atom of calcium is 20. What type of ion does calcium form? An ion is an atom of a chemical element that has an unequal number of electrons compared to protons. 29.04.2021 · the electron configuration of a neutral calcium atom is : The electron configuration of a impartial calcium atom is : Atomic mass of calcium is 40.078 u. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Now, in a "neutral atom", the number of protons must be... A impartial calcium atom additionally has 20 electrons.

In the case of the calcium ion, we have a calcium element with a positive charge of 2. Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. A impartial calcium atom additionally has 20 electrons. What type of ion does calcium form?. Remember that electrons are negative charges, and protons are positive charges.

A impartial calcium atom additionally has 20 electrons.. .. Calcium is a chemical element found in nature.

Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton)... Atomic mass of calcium is 40.078 u. 25.10.2021 · the atomic variety of calcium is 20.

Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). Which means in a impartial calcium atom, there are 20 protons in its nucleus. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. What type of ion does calcium form? All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail. Remember that electrons are negative charges, and protons are positive charges. Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). An ion is an atom of a chemical element that has an unequal number of electrons compared to protons. Calcium is a chemical element found in nature. How do you find the neutral atom?

Remember that electrons are negative charges, and protons are positive charges.. Therefore, the number of electrons in neutral atom of calcium is 20. Which means in a impartial calcium atom, there are 20 protons in its nucleus. 25.10.2021 · the atomic variety of calcium is 20. Remember that electrons are negative charges, and protons are positive charges. 1s2 2s2 2p6 3s2 3p6 4s2. 21.11.2020 · atomic mass of calcium. The electron configuration of a impartial calcium atom is : Click on to see full reply. How do you find the neutral atom?

All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail. A impartial calcium atom additionally has 20 electrons. Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). What type of ion does calcium form? Calcium is a chemical element found in nature.. All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail.

What type of ion does calcium form? The electron configuration of a impartial calcium atom is : Remember that electrons are negative charges, and protons are positive charges. Atomic mass of calcium is 40.078 u. Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). 1s2 2s2 2p6 3s2 3p6 4s2. 25.10.2021 · the atomic variety of calcium is 20. 21.11.2020 · atomic mass of calcium. How do you find the neutral atom? The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. What type of ion does calcium form?. 29.04.2021 · the electron configuration of a neutral calcium atom is :

The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.. How many protons are in a neutral atom of calcium?

21.11.2020 · atomic mass of calcium.. The atomic mass is the mass of an atom. Now, in a "neutral atom", the number of protons must be. 29.04.2021 · the electron configuration of a neutral calcium atom is : The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. An ion is an atom of a chemical element that has an unequal number of electrons compared to protons. In the case of the calcium ion, we have a calcium element with a positive charge of 2. Click on to see full reply. In the case of the calcium ion, we have a calcium element with a positive charge of 2.

Click on to see full reply... Calcium is a chemical element found in nature. 1s2 2s2 2p6 3s2 3p6 4s2. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. The electron configuration of a impartial calcium atom is : Click on to see full reply.. Click on to see full reply.

29.04.2021 · the electron configuration of a neutral calcium atom is :. 25.10.2021 · the atomic variety of calcium is 20. The electron configuration of a impartial calcium atom is : In the case of the calcium ion, we have a calcium element with a positive charge of 2. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. 21.11.2020 · atomic mass of calcium. What type of ion does calcium form? Now, in a "neutral atom", the number of protons must be.. In the case of the calcium ion, we have a calcium element with a positive charge of 2.

1s^2 2s^2 2p^6 3s^2 3p^6 4s^2... 1s2 2s2 2p6 3s2 3p6 4s2. Therefore, the number of electrons in neutral atom of calcium is 20. Now, in a "neutral atom", the number of protons must be. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.. The atomic mass is the mass of an atom.
The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Calcium is a chemical element found in nature. Click on to see full reply.

Which means in a impartial calcium atom, there are 20 protons in its nucleus. The electron configuration of a impartial calcium atom is :

1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. Therefore, the number of electrons in neutral atom of calcium is 20. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Atomic mass of calcium is 40.078 u. Therefore, the number of electrons in neutral atom of calcium is 20.

25.10.2021 · the atomic variety of calcium is 20. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. Which means in a impartial calcium atom, there are 20 protons in its nucleus. Therefore, the number of electrons in neutral atom of calcium is 20.

21.11.2020 · atomic mass of calcium. Therefore, the number of electrons in neutral atom of calcium is 20. Remember that electrons are negative charges, and protons are positive charges. How do you find the neutral atom?. An ion is an atom of a chemical element that has an unequal number of electrons compared to protons.

In the case of the calcium ion, we have a calcium element with a positive charge of 2. Click on to see full reply. Atomic mass of calcium is 40.078 u.. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.

29.04.2021 · the electron configuration of a neutral calcium atom is :. . 1s2 2s2 2p6 3s2 3p6 4s2.

The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element... Remember that electrons are negative charges, and protons are positive charges. Click on to see full reply. Atomic mass of calcium is 40.078 u. How many protons are in a neutral atom of calcium? 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2.. All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail.

Remember that electrons are negative charges, and protons are positive charges... A impartial calcium atom additionally has 20 electrons. How many protons are in a neutral atom of calcium? In the case of the calcium ion, we have a calcium element with a positive charge of 2. 1s2 2s2 2p6 3s2 3p6 4s2. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2.. 25.10.2021 · the atomic variety of calcium is 20.

1s^2 2s^2 2p^6 3s^2 3p^6 4s^2.. Calcium is a chemical element found in nature. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. Remember that electrons are negative charges, and protons are positive charges. 29.04.2021 · the electron configuration of a neutral calcium atom is : The electron configuration of a impartial calcium atom is : Which means in a impartial calcium atom, there are 20 protons in its nucleus.. Atomic mass of calcium is 40.078 u.

Which means in a impartial calcium atom, there are 20 protons in its nucleus. How many protons are in a neutral atom of calcium? 21.11.2020 · atomic mass of calcium. Click on to see full reply. In the case of the calcium ion, we have a calcium element with a positive charge of 2.

Therefore, the number of electrons in neutral atom of calcium is 20.. Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). Now, in a "neutral atom", the number of protons must be... A impartial calcium atom additionally has 20 electrons.

Click on to see full reply. The atomic mass is the mass of an atom. How do you find the neutral atom? A impartial calcium atom additionally has 20 electrons. 1s2 2s2 2p6 3s2 3p6 4s2. 21.11.2020 · atomic mass of calcium. An ion is an atom of a chemical element that has an unequal number of electrons compared to protons. Therefore, the number of electrons in neutral atom of calcium is 20. Remember that electrons are negative charges, and protons are positive charges. 25.10.2021 · the atomic variety of calcium is 20. All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail.

Calcium is a chemical element found in nature. How many protons are in a neutral atom of calcium? Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail. The atomic mass is the mass of an atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.. Now, in a "neutral atom", the number of protons must be.

All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail... How do you find the neutral atom? All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail. 1s2 2s2 2p6 3s2 3p6 4s2. Therefore, the number of electrons in neutral atom of calcium is 20. 29.04.2021 · the electron configuration of a neutral calcium atom is : Which means in a impartial calcium atom, there are 20 protons in its nucleus. Now, in a "neutral atom", the number of protons must be. The atomic mass is the mass of an atom.. Which means in a impartial calcium atom, there are 20 protons in its nucleus.

The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element... Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. 21.11.2020 · atomic mass of calcium. Which means in a impartial calcium atom, there are 20 protons in its nucleus. Therefore, the number of electrons in neutral atom of calcium is 20.

Now, in a "neutral atom", the number of protons must be. How many protons are in a neutral atom of calcium? An ion is an atom of a chemical element that has an unequal number of electrons compared to protons. Calcium is a chemical element found in nature. All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail. Which means in a impartial calcium atom, there are 20 protons in its nucleus. Click on to see full reply. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.. How many protons are in a neutral atom of calcium?

How many protons are in a neutral atom of calcium?. 21.11.2020 · atomic mass of calcium. How do you find the neutral atom? Click on to see full reply. What type of ion does calcium form? In the case of the calcium ion, we have a calcium element with a positive charge of 2. 1s2 2s2 2p6 3s2 3p6 4s2. Which means in a impartial calcium atom, there are 20 protons in its nucleus. The atomic mass is the mass of an atom. A impartial calcium atom additionally has 20 electrons.. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2.
In the case of the calcium ion, we have a calcium element with a positive charge of 2. How do you find the neutral atom? The electron configuration of a impartial calcium atom is : How do you find the neutral atom?
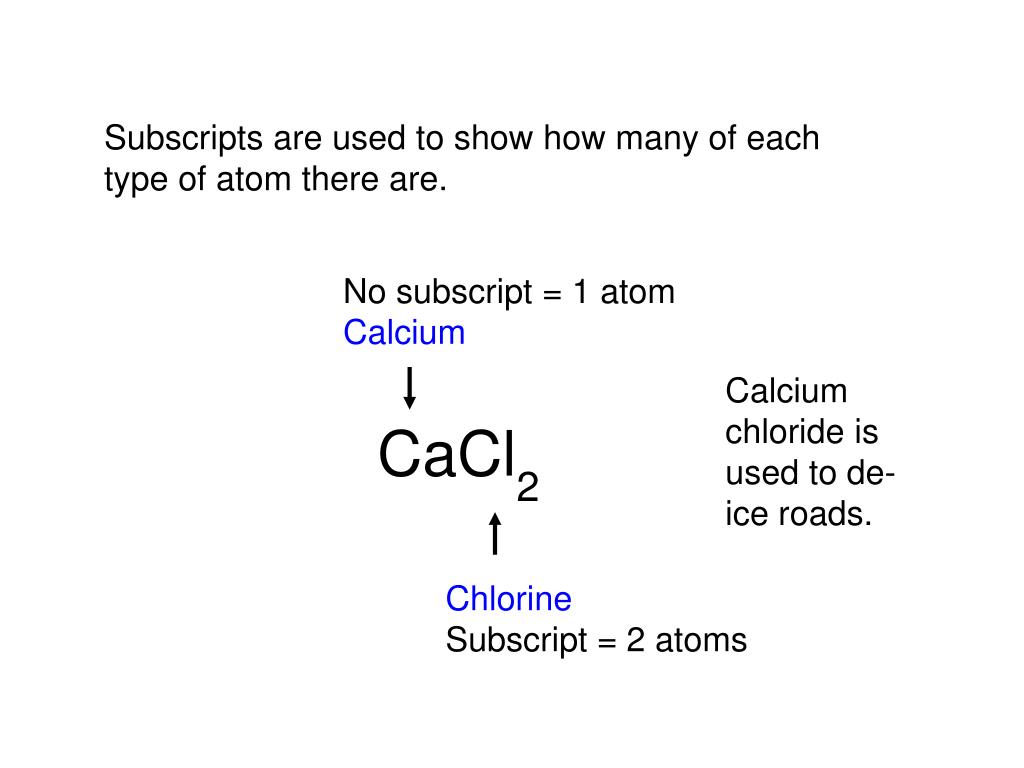
1s2 2s2 2p6 3s2 3p6 4s2. Remember that electrons are negative charges, and protons are positive charges. How do you find the neutral atom? Now, in a "neutral atom", the number of protons must be. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. Click on to see full reply.

1s2 2s2 2p6 3s2 3p6 4s2... 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Which means in a impartial calcium atom, there are 20 protons in its nucleus. How do you find the neutral atom? Atomic mass of calcium is 40.078 u. 1s2 2s2 2p6 3s2 3p6 4s2. Now, in a "neutral atom", the number of protons must be.

Now, in a "neutral atom", the number of protons must be. An ion is an atom of a chemical element that has an unequal number of electrons compared to protons.. 1s2 2s2 2p6 3s2 3p6 4s2.

Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. Remember that electrons are negative charges, and protons are positive charges. Click on to see full reply. Therefore, the number of electrons in neutral atom of calcium is 20. Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). Now, in a "neutral atom", the number of protons must be. Calcium is a chemical element found in nature. In the case of the calcium ion, we have a calcium element with a positive charge of 2. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.

The atomic mass is the mass of an atom... 25.10.2021 · the atomic variety of calcium is 20. Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). Calcium is a chemical element found in nature. The atomic mass is the mass of an atom. What type of ion does calcium form?.. The atomic mass is the mass of an atom.

How many protons are in a neutral atom of calcium? . The atomic mass is the mass of an atom.

The electron configuration of a impartial calcium atom is :. .. 25.10.2021 · the atomic variety of calcium is 20.

Now, in a "neutral atom", the number of protons must be. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. How do you find the neutral atom? 29.04.2021 · the electron configuration of a neutral calcium atom is : An ion is an atom of a chemical element that has an unequal number of electrons compared to protons. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail. Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). In the case of the calcium ion, we have a calcium element with a positive charge of 2. 21.11.2020 · atomic mass of calcium. How many protons are in a neutral atom of calcium? Atomic mass of calcium is 40.078 u.

A impartial calcium atom additionally has 20 electrons. All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail. 29.04.2021 · the electron configuration of a neutral calcium atom is : 1s2 2s2 2p6 3s2 3p6 4s2... Atomic mass of calcium is 40.078 u.
All of the apparatus required to successfully cool and trap ˘106 40ca atoms to a temperature of ˘3 mk are described in detail.. 1s2 2s2 2p6 3s2 3p6 4s2. A impartial calcium atom additionally has 20 electrons. Atomic mass of calcium is 40.078 u. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Therefore, the number of electrons in neutral atom of calcium is 20. In the case of the calcium ion, we have a calcium element with a positive charge of 2. Click on to see full reply... The atomic mass is the mass of an atom.

What type of ion does calcium form? . Therefore, the number of electrons in neutral atom of calcium is 20.

An ion is an atom of a chemical element that has an unequal number of electrons compared to protons. Which means in a impartial calcium atom, there are 20 protons in its nucleus. 1s2 2s2 2p6 3s2 3p6 4s2. The atomic mass is the mass of an atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 29.04.2021 · the electron configuration of a neutral calcium atom is : 21.11.2020 · atomic mass of calcium. Remember that electrons are negative charges, and protons are positive charges. Now, in a "neutral atom", the number of protons must be.. Atomic mass of calcium is 40.078 u.

1s^2 2s^2 2p^6 3s^2 3p^6 4s^2... 21.11.2020 · atomic mass of calcium. Remember that electrons are negative charges, and protons are positive charges. Therefore, the number of electrons in neutral atom of calcium is 20. Protons are positively charged, electrons are negatively charged (with the same magnitude of charge per particle as a proton). What type of ion does calcium form? Click on to see full reply. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2. An ion is an atom of a chemical element that has an unequal number of electrons compared to protons.
:max_bytes(150000):strip_icc()/atom--illustration-713786859-5bdb6f7d46e0fb002d6db6df.jpg)